CagriSema vs Tirzepatide: The 22.7% Headline (and the Asterisk Nobody Mentions)
TL;DR: Novo Nordisk's CagriSema is real, it's filed with the FDA, and it produced 22.7% weight loss in the headline REDEFINE 1 trial — bigger than Wegovy. But in the only head-to-head comparison against Mounjaro/Zepbound's tirzepatide (REDEFINE 4, February 2026), CagriSema lost — 23.0% vs 25.5% — and missed its noninferiority endpoint. If you're already on tirzepatide and tolerating it well, there's no scientific reason to wait for CagriSema.
The drug everyone's talking about
CagriSema is a once-weekly injection that combines two molecules: semaglutide (the same GLP-1 agonist in Wegovy and Ozempic) and cagrilintide (a long-acting analog of amylin, a satiety hormone made by the same pancreatic beta cells as insulin). Novo Nordisk filed CagriSema's New Drug Application with the FDA on December 18, 2025, with a decision expected late 2026 (PRNewswire, Dec 18 2025).
For Novo, CagriSema is the answer to a problem the company invented and then lost: Mounjaro and Zepbound (Eli Lilly's tirzepatide) deliver more weight loss than Wegovy, and US patients have been switching. CagriSema was designed to close that gap.
It worked. But not by enough.
REDEFINE 1: the 22.7% that made headlines
Published in the New England Journal of Medicine in June 2025, REDEFINE 1 randomized 3,417 adults with obesity (no diabetes) to one of four arms for 68 weeks: CagriSema 2.4 mg / 2.4 mg, semaglutide 2.4 mg alone, cagrilintide 2.4 mg alone, or placebo.
The headline result (DOI: 10.1056/NEJMoa2502081):
| Treatment | Mean weight loss at 68 weeks |
|---|---|
| CagriSema | −22.7% (efficacy estimand) / −20.4% (treatment-policy) |
| Semaglutide alone | −14.9% |
| Cagrilintide alone | −11.5% |
| Placebo | −3.0% |
Half the CagriSema arm (50.7%) dropped below a BMI of 30 by trial end — they moved out of the "obese" category. That's the Novo press release line. It's a real number from a 3,417-person Phase 3 trial published in NEJM. It is also the only number most coverage mentioned.
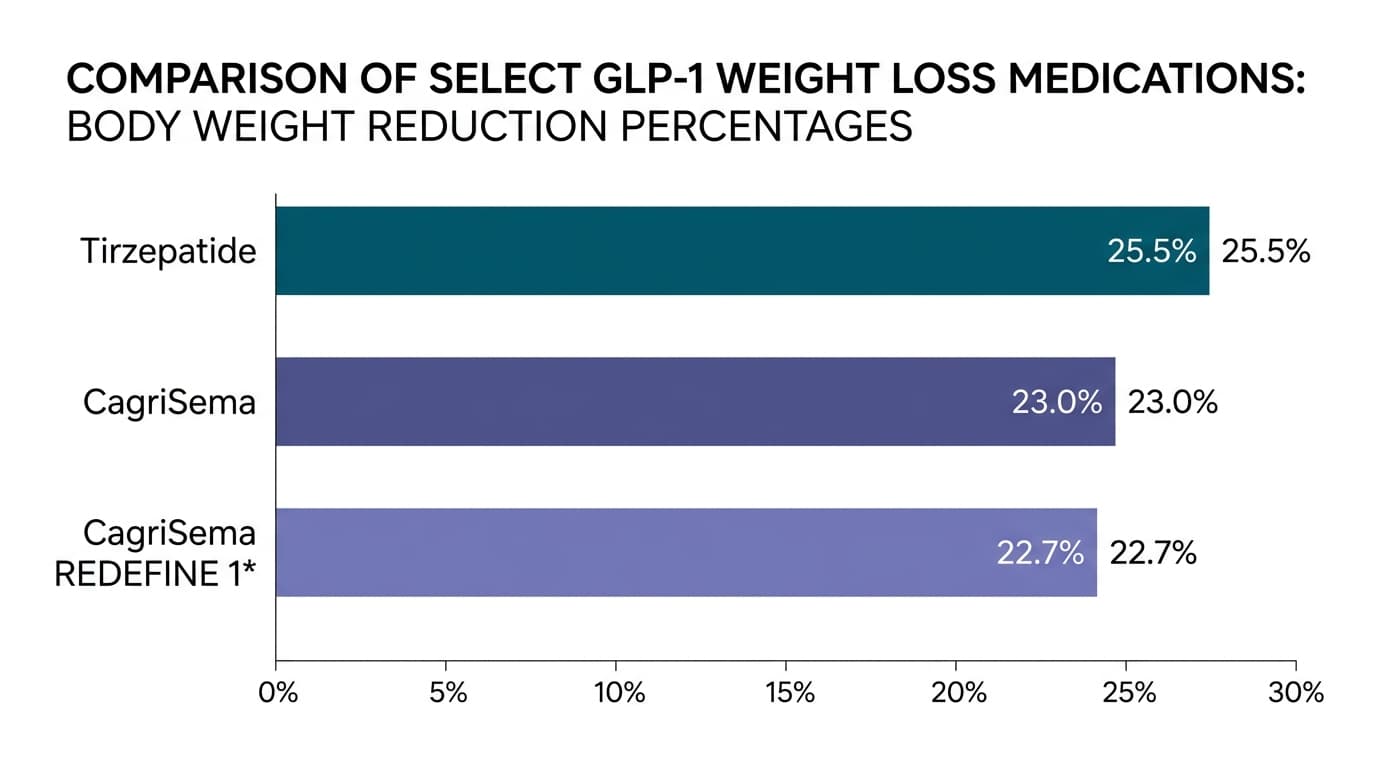
REDEFINE 4: the head-to-head that didn't go Novo's way
In February 2026, Novo published REDEFINE 4 — the head-to-head trial that pitted CagriSema directly against tirzepatide 15 mg in 809 adults with obesity over 84 weeks (Novo press release, Feb 23 2026).
The results:
| Endpoint | CagriSema 2.4/2.4 mg | Tirzepatide 15 mg |
|---|---|---|
| Weight loss (efficacy) | −23.0% | −25.5% |
| Weight loss (treatment-regimen) | −20.2% | −23.6% |
| % reaching highest dose | 57.3% | 82.5% |
The primary endpoint of noninferiority was not met. That is unusual to state plainly in pharma communications, and Novo did. Tirzepatide produced more weight loss than CagriSema and was better tolerated at the top dose. In the only direct comparison that exists, the drug you can already get today is more effective than the drug Novo is asking the FDA to approve.
What the diabetes trial showed
REDEFINE 2, in 1,206 adults with type 2 diabetes (PMID 40544432), showed CagriSema produced 13.7% weight loss vs placebo's 3.4% over 68 weeks. 73.5% of CagriSema patients reached HbA1c ≤6.5% vs 15.9% on placebo. A separate active-controlled T2D trial, REIMAGINE 2 (Feb 2026), beat semaglutide 2.4 mg head-to-head on both weight loss (−14.2% vs −10.2%) and HbA1c reduction (−1.91 vs lower).
So if you have type 2 diabetes, CagriSema vs Ozempic is a clean win. But "stronger than Ozempic" was never the question — the question was always "stronger than Mounjaro." And the answer is no.
Why combine amylin with GLP-1 in the first place?
Cagrilintide is what Novo's chemists call an amylin analog. Amylin is the second hormone your pancreatic beta cells secrete alongside insulin — it slows gastric emptying, suppresses postprandial glucagon, and signals satiety through the brainstem (J Med Chem, 2021). It works through a different receptor system than GLP-1.
The theory was elegant: stacking two satiety pathways should be more than additive. In the REDEFINE 1 data, semaglutide alone delivered 14.9% weight loss; adding cagrilintide pushed it to 20.4%. The amylin component contributed a real 5.5 percentage points.
The problem is that tirzepatide's bet — GLP-1 plus GIP (a different gut hormone) — turned out to be even more powerful, and Lilly got there first. Pharmacology is a tournament. Coming in second still puts you in the conversation, but it doesn't put you on the prescription pad.
Side effects: not gentler
There was hope that adding amylin to GLP-1 might soften the GI side effects that drive about a third of GLP-1 patients off their medication in year one. The data doesn't support that hope.
CagriSema's side effect profile in REDEFINE 1:
- Nausea: ~55% (vs 12.6% placebo)
- Vomiting: ~26%
- Constipation: ~31%
- Total GI adverse events: 79.6% (vs 39.9% placebo)
This is roughly the same as semaglutide alone. Stacking cagrilintide on top of sema doesn't make either drug gentler — it just adds satiety effect on top of satiety effect.
Should you wait for CagriSema?
Three patient profiles, three different answers:
1. You're on tirzepatide (Mounjaro or Zepbound) and tolerating it. No. The only head-to-head says tirzepatide delivers more weight loss and is easier to titrate. Stay where you are.
2. You're on semaglutide (Wegovy or Ozempic) and plateaued. Maybe. The mechanism gain over sema alone is real (~5.5 percentage points). But your simpler move is the conversation about switching to tirzepatide — which is already approved, already covered for many plans, and has the head-to-head win.
3. You have type 2 diabetes on metformin and haven't started a GLP-1 yet. CagriSema's REIMAGINE 2 data on HbA1c reduction is genuinely best-in-class. If you can wait until late 2026, it's worth the conversation. If you can't wait, tirzepatide is the bridge.
What this means for the pipeline
CagriSema's REDEFINE 4 result is a moment for the whole field. It says GLP-1 + amylin combinations have a ceiling that's still below GLP-1 + GIP. It says the next leap is going to come from triple agonists (retatrutide's GLP-1 + GIP + glucagon) or something we haven't seen yet — not from polishing existing single-hormone combinations.
For the patient looking at the news cycle: the era of every-new-drug-is-better is over. Tirzepatide is the current ceiling, and the drugs after it have to actually beat it in a head-to-head, not just produce a big number in a placebo-controlled trial.
Key Takeaways
- CagriSema produced 22.7% weight loss in REDEFINE 1 vs 14.9% for semaglutide alone — the amylin component adds ~5.5 percentage points.
- CagriSema LOST to tirzepatide in REDEFINE 4 (23.0% vs 25.5%) and missed its noninferiority endpoint. Tirzepatide was also easier to titrate (82.5% reached top dose vs 57.3%).
- NDA filed Dec 18, 2025; FDA decision expected late 2026.
- Side effects are not gentler than semaglutide alone — nausea 55%, GI events 79.6%.
- If you're on tirzepatide and tolerating it, there's no scientific reason to wait for CagriSema. If you have T2D and want best-in-class HbA1c reduction, CagriSema is worth the conversation.
Build the habits that outlast any drug switch
Whichever GLP-1 you end up on — current, next, or pipeline — the medication is the easier half. The harder half is the daily structure: protein at every meal, the resistance-training session you don't skip, the hydration habit, the sleep window. That is what makes the weight loss durable when the prescription changes (and it will change). Gila is built to do that work alongside you, regardless of which molecule is in the pen.
Frequently asked questions
Is CagriSema better than Mounjaro for weight loss? No. In REDEFINE 4 (open-label, head-to-head, n=809), tirzepatide produced 25.5% weight loss vs CagriSema's 23.0% over 84 weeks, and was better tolerated at the highest dose.
When will CagriSema be FDA-approved? Novo Nordisk filed the NDA on December 18, 2025. FDA decisions on new molecules typically take 10-12 months, so a late-2026 decision is expected, with US market availability likely in early 2027 if approved.
What are CagriSema's side effects? The most common are nausea (~55%), constipation (~31%), and vomiting (~26%). Total GI adverse events were 79.6% in REDEFINE 1 versus 39.9% on placebo. The profile is roughly the same as semaglutide alone; adding cagrilintide did not soften the GI burden.
How is CagriSema different from Wegovy? Same semaglutide base, plus cagrilintide — a long-acting amylin analog that targets a separate satiety pathway in the brainstem and gut. The combination adds ~5.5 percentage points of weight loss over semaglutide alone in head-to-head data.
Will CagriSema replace my Wegovy prescription? Unlikely as a direct 1:1 swap once approved. It produces more weight loss than semaglutide alone but with the same dosing burden, similar side effects, and (importantly) without beating tirzepatide. Your prescriber's call will likely depend on coverage and your specific weight-loss plateau.
Is CagriSema "stronger Ozempic"? Functionally yes — it's semaglutide plus a second satiety hormone analog. But it's a combination product, not a different drug class. Calling it "stronger Ozempic" is fair shorthand; calling it a "next-generation GLP-1" overstates the mechanism leap.
Sources
- Garvey et al. CagriSema vs Placebo in Adults with Obesity. NEJM, June 22 2025. DOI: 10.1056/NEJMoa2502081
- CagriSema in Type 2 Diabetes (REDEFINE 2). NEJM, Aug 14 2025. PMID: 40544432
- Novo Nordisk. CagriSema vs Tirzepatide in REDEFINE 4. Press release, Feb 23 2026
- Novo Nordisk. REIMAGINE 2 Topline. Press release, Feb 2 2026
- Novo Nordisk. FDA NDA Filing. PRNewswire, Dec 18 2025
- Kruse T et al. The Discovery of Cagrilintide. J Med Chem. DOI: 10.1021/acs.jmedchem.1c00565
More from Gila
Ready to start your GLP-1 journey?
Gila helps you build lasting habits, understand your body, and stay on track. Join the pilot for free.
Join the Pilot
